Dyno Frontiers Network
Connecting gene therapy developers to delivery technology and trusted partners to demonstrate the power of innovative genetic payloads.

Announcing the Dyno Frontiers Network
As genetic technologies advance, gene therapy developers increasingly need high-performance AAV delivery vectors to realize the therapeutic potential of their innovative payload strategies. Many gene therapy developers lack access to advanced AAV delivery technology, and even when developers can evaluate new capsids, a lack of standardized methods for quantifying capsid performance creates uncertainty about whether a novel payload strategy will be effective with subsequent generations of capsids. Additionally, some emerging payload strategies require distinct evaluation methods and data-types which generic capsid evaluation metrics don't address – a challenge which requires additional developer resources and time to overcome. Dyno addressed this barrier with the launch of Frontiers in 2025, enabling gene therapy developers to increase confidence in the therapeutic potential of their innovative payloads.
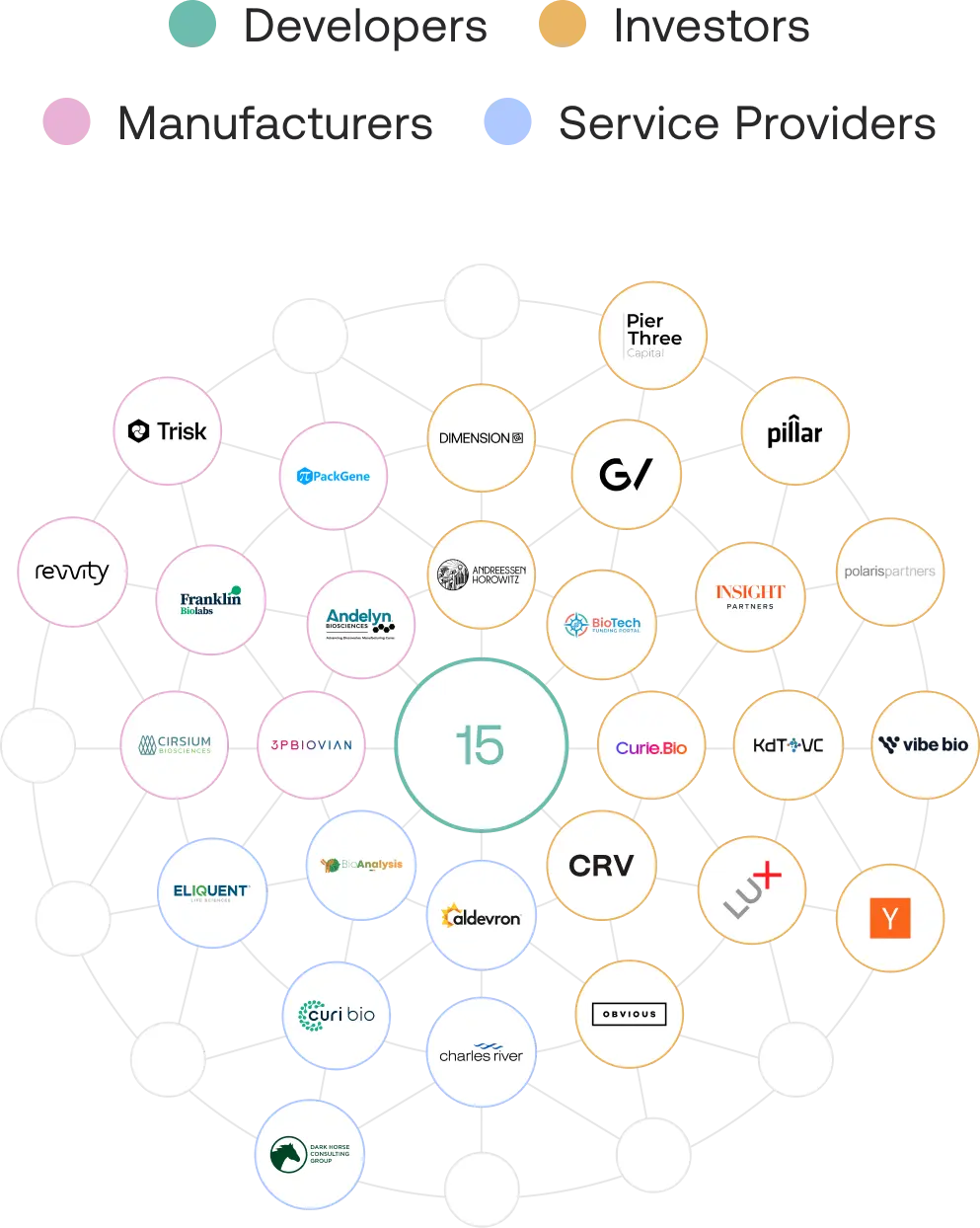
With delivery addressed, the next challenge is manufacturing. Developers must individually vet and select among many manufacturers, then enter new relationships where the capsid, the manufacturing process, and the payload are all unfamiliar to each other – creating significant execution risk. When setbacks occur, it becomes difficult to isolate the source of the issue, leading to costly troubleshooting and delays that ultimately cost patients the most. Dyno addressed this barrier by establishing an integrated manufacturing marketplace of partners who have proven they can produce Dyno® capsids within their established processes, giving developers a choice of manufacturers experienced with Dyno's capsids, reducing uncertainty in yield, cost, and overall success, while accelerating progress to the clinic.
With manufacturing addressed, the questions developers asked shifted towards wanting to understand who are capable development partners to help them generate preclinical data and strategize on early regulatory interactions. Identifying, evaluating, and onboarding these service providers takes time developers can't afford to lose, and picking the wrong partner can set a program back months. To give developers choice and a stronger starting point, Dyno integrated a select group of regulatory consultants and preclinical CROs experienced in AAV therapeutic development.
With execution foundations in place, developers still face the challenge of reaching investors prepared to focus on what's truly differentiated about their payload. To make this easier for developers, Dyno proactively curated a group of investors already knowledgeable about Dyno and Frontiers, where Dyno provides developers warm introductions to potential seed and Series A financing partners. While there is no obligation for these investors to fund any individual developer, being part of Frontiers signals to investors that a developer is building on a strong foundation, and strengthens the pitch for companies considering startup accelerators such as Y Combinator.
By joining the Dyno Frontiers Network, developers gain:
- No-cost, non-exclusive research access to high-performance Dyno capsids, optimized for CNS, muscle, eye.
- Advisory support from Dyno on capsid selection, study design, and the performance metrics that matter for novel payload strategies.
- Access to Dyno curated capsid performance data insights across diverse payload modalities, helping to predict performance and anticipate what to expect from next-generation capsids.
- Trusted manufacturing partners with demonstrated experience producing Dyno capsids.
- CROs and regulatory consultants with expertise in AAV gene therapy and Dyno's platform.
- Access to investors in early-stage gene therapy who have already done diligence on Dyno's platform and Frontiers.
The Dyno Frontiers Network reflects Dyno's partner-centric model – a curated ecosystem where collaboration and competition coexist among trusted, capable partners, giving gene therapy developers greater choice in bringing successful medicines to patients more easily.
If you are a gene therapy developer advancing an innovative payload, a manufacturer or service provider with something valuable and differentiated to offer developers, or an investor focused on early-stage gene therapy, we’d welcome the opportunity to partner together. Reach out to partnerwith@dynotx.com.
.webp)